Bard PowerPort Lawsuit
Hundreds of adverse events associated with Bard PowerPorts have been reported to the FDA, and lawsuits are being filed. If you have been injured after being implanted with a Bard PowerPort device, you may have a claim. Contact The Lanier Law Firm at 800-723-3216 today for a free case evaluation.
Home » Product Liability Litigation » Bard PowerPort Lawsuit
- Legally Reviewed By:
- Rebecca Phillips
- Page Last Updated:
- April 27, 2026
Navigate This Page
- Bard PowerPort Litigation Updates and Timeline
- Why Choose the Lanier Law Firm to Handle Your Bard PowerPort Lawsuit?
- What Is a Bard PowerPort?
- What Is the Problem with Bard PowerPorts?
- The Effects and Complications Associated with Bard PowerPort Catheters
- Liability for Defective PowerPort Catheter Defects
- When Should I Contact a Bard PowerPort Attorney?
- Contact the Lanier Law Firm to Start Your Claim Today
- Bard Port Lawsuit FAQs
The Lanier Law Firm is actively pursuing claims against Bard PowerPort for defective medical devices that have caused severe health complications.
Bard PowerPort is a medical device implanted surgically in the human body to provide direct access to veins for delivering medications and fluids.
The issue arises from the catheter, which contains barium sulfate that has been shown to degrade polyurethane and silicone when in contact with human tissue, leading to complications such as thrombosis, myocardial infarction symptoms, pulmonary embolism, organ damage, infections, and more.
The FDA has received hundreds of adverse event reports from patients implanted with Bard PowerPorts, but the company continues to market the products as safe and effective without recalling them, adjusting the product, or adding warnings to the label.
Contact us now for a free consultation if you or a loved one have been implanted with a Bard PowerPort device and have experienced serious complications like blood clots, heart-related symptoms, infections, or device failure.
Bard PowerPort Litigation Updates and Timeline
Transcript:
Jon Robinson:
You’re in leadership on Bard, right? What was it about that case that checked the boxes for you? You’re like, this is the one that we really should be investing time, money, and resources in.
Rebecca Phillips:
Yeah, I’m one of the co-leads. There’s 3 co-leads in this case, and I’m one of the co-leads. It’s my first experience being a co-lead in a litigation. There’s a lot to learn.
We’ve got about 20 or 25 firms participating in this. And you know, knock on wood, so far so good. I think we’re doing all right.
There’s about 2000 cases filed. We’re about to get the case specific briefing behind us and we’re about to go to trial in 2026.
Jon Robinson:
So the first bellwether.
Rebecca Phillips:
The first bellwether, well actually six bellwethers will happen in 2026.
Yeah, so if you’re not familiar with this tort, the signature injuries are a fracture of the catheter, which can usually go into the patient’s heart. If it embolizes, it’ll go into the patient’s heart, do some damage, they would have to have it removed. Scary situation.
The catheter can get infected, and when it does get infected, it’s a bloodstream infection. At the very minimum, it requires removal of the port, which the patient needs for chemotherapy, typically.
Jon Robinson:
It is because it’s supposed to be close to their heart so that medicine can circulate through the blood, right?
Rebecca Phillips:
Yeah. So, so there’s that, or the port can thrombose, and when you get thrombosis that embolizes, you end up with pulmonary embolism, also life threatening. So, these injuries from these ports can be very serious at the time in the patient’s life when they do not need to be dealing with serious complications.
And, the most remarkable thing about this case to me is that since at least, and this is being generous, but since at least the early 2010s, there has been a solution to these problems, and Bard and Becton Dickinson never implemented the solutions to those problems.
It’s really, in my opinion, a factor, just the fact that they control, since about 2006, they control 70 to 75% of the port market and if you just limit that to the United States, it’s even more. They just don’t have an economic incentive to make these ports better.
April 6, 2026: Soon First Bellwether Trial, Plaintiff Septic After Port Implant
The first bellwether Plaintiff has survived summary judgment. A pretrial conference is set for April 9, and the parties will go to trial on April 21. The case concerns an infection injury, where the Plaintiff went septic ten days after port implant.
February 3, 2026: Court Excludes Defendants’ Expert Opinions
The parties have concluded dispositive, case-specific motion briefing for five bellwether cases, and the Court has begun to rule on case-specific motions for the first bellwether trial, an infection injury. Most notably, the Court has excluded several of the opinions of Defendants’ experts who claim that the infection was not caused by Defendants’ defective port. Those rulings help to narrow Defendants’ available defenses for trial.
January 14, 2026: Daubert and MSJ Briefings Submitted
The parties have submitted all Daubert and MSJ briefings on general causation and on 5 of 6 bellwether cases, and Defendants have moved to seal most of that briefing. Judge Campbell will begin ruling on all of those motions, including the motions to seal, on a rolling basis. The first trial, a short-onset infection case, remains on schedule to begin in April 2026.
September 17, 2025: Trial Dates Set in Implanted Port Catheter MDL
Over 2,000 cases are now on file in the implanted port catheter multidistrict litigation in the U.S. District Court of Arizona. More cases are also being filed in New Jersey and in state court in Arizona. In Arizona, the first trial has been set for August 2026, and in the federal MDL, six trials are scheduled to begin in March 2026 and to run through the end of the year.
The 2026 trials will focus on injuries caused by infection, thrombosis, and fracture. Lanier Law Firm founder Mark Lanier plans to try one of the bellwether cases.
The parties are currently engaged in motion practice. Defendants elected not to file common-issue motions for summary judgment, but both sides filed numerous expert challenges. Case-specific motions, focused specifically on bellwether trials, will be filed in October.
August 6, 2025: Bard PowerPort Co-Lead Counsel on Expert Reports and Motions
Rebecca Phillips, Co-Lead Counsel on the Bard PowerPort litigation, provided the following: “On August 4, Plaintiffs served case-specific expert reports for bellwether cases. Common-issue expert depositions and case-specific fact witness depositions remain ongoing. Motions for summary judgment and expert challenges are expected later this month.”
May 14, 2025: Court Selects Bellwether Cases for 2026 Bard PowerPort Trial
The Court has selected six bellwether cases for trial in 2026. The Court also selected one alternate case, should one of the other six cases be resolved before trial. Of the seven cases, three are infection injuries, two are thrombus/occlusion injuries, and two, including the alternate, are fracture injuries. This selection will be followed by a short period of additional discovery and submission of case-specific expert reports, and briefing on summary judgment and expert challenges begins in the fall.
May 7, 2025: Bellwether Group 1 Discovery Closed
General and case-specific discovery for Bellwether Group 1 has closed, and the parties will submit briefing today about which six cases should move forward as bellwethers. After the Court decides that issue, additional case-specific discovery may continue. The parties are also in the process of exchanging expert reports, rebuttals, and beginning expert depositions.
April 9, 2025: Expert Reports on Port Catheter Defects
Plaintiffs served their general expert reports on March 14, 2025. Plaintiffs disclosed a dozen experts who support Plaintiffs’ claims that Defendants’ port catheters are defective: Michael Beatrice, PhD; Bernard Camins, MD; Ahmed El-Ghannam, PhD; Darren Hurst, MD; William Jarvis, MD, Madris Kinard, MBA; Deborah Leckband PhD; Brian McVerry, PhD; Buddy Ratner, PhD; Amir Sheikhi, PhD; Becky Smith, MD; and Jeffrey Weinstein, MD.
The parties have completed general discovery and are winding up case-specific discovery for bellwether selection. At the last status conference, per Defendants’ request, the Court moved the deadline for the parties to agree on six bellwether cases or to submit opposing briefing about representative cases to April 28. The next conference is set for May 1.
January 16, 2025: Bard PowerPort Bellwether Depositions
Discovery continues, and depositions are ongoing. This month, depositions of 15 potential bellwether plaintiffs will begin, followed soon after by treating physicians. These depositions are intended to help the parties narrow the bellwether pool from 15 cases down to 6 cases that will be tried in early 2026.
In December, the parties could not reach agreement over topics for corporate representative deposition, and the Court issued an order to govern the issue. The next hearing is set for today, January 16, 2025, and the parties are expected to argue over Defendants’ withholding of non-privileged evidence. Plaintiffs have already been successful in getting Defendants to release such evidence through the course of earlier disputes.
October 17, 2024: Court Orders Further Briefing on Bard PowerPort Discovery Issues
The Court held a hearing on October 3 to discuss the progress of discovery. The parties argued regarding Defendants’ claimed privileges and redactions of evidence and regarding a potential, short extension of the discovery schedule due to Defendants late production of documents and continual moving back of witness deposition dates. The Court ordered more briefing on the issues. The parties also argued about evidence regarding the materials out of which Defendants’ catheters are made, and the Court determined that, if in the future Plaintiffs discovery that Defendants have withheld information, they may re-approach the Court regarding an appropriate sanction.
Meanwhile, depositions continue. Co-lead counsel, Lanier Law Firm attorney Rebecca Phillips, said that, while witness testimony cannot yet be made public, “Our team is doing good work in there.”
September 16, 2024: Objections to Successor Liability Discovery Rejected in Bard Litigation
Discovery continues, and depositions of Defendants’ witnesses have begun. The Court held a conference with the parties on August 16, 2024, to discuss the status of the case and ongoing discovery issues. The biggest issue was that, from the start of discovery, Defendants have opposed Plaintiffs’ discovery into the corporate relationship between Bard and BD for the purpose of proving successor liability. The Court – for a second time – rejected the Defendants argument. Unless the parties are able to reach an agreed stipulation on the relationship between Bard and BD, Plaintiffs’ successor liability discovery will continue.
Another hearing is scheduled for October, and at the last status conference, our own Rebecca Phillips who is on the leadership team for this litigation, told the Court that it may anticipate an argument about evidence that Defendants are withholding as privileged.
June 11, 2024: Status Conference Held, Depositions Set for August
The Court held another status conference with the parties on May 24, 2024 to receive an update on continued discovery issues. At present, the parties remain on track with the production of evidence so that depositions can begin in August of 2024. According to co-lead counsel, Rebecca Phillips, an attorney at the Lanier Law Firm, “It is important to get these cases to trial as quickly as we reasonably can. Since many of our clients are very ill, they can’t see justice soon enough.”
May 7, 2024: MDL Court to Address Bard Discovery Progress
The parties are scheduled to appear before the MDL Court again on May 10 to discuss ongoing discovery issues, including whether the parties are on track to meet current discovery deadlines. At present, the parties continue to engage in the discovery process of identifying relevant witnesses and documents for production. Depositions of those witnesses are still scheduled to begin in August of this year.
April 22, 2024: Bard PowerPort Bellwether Pool Closed
The bellwether pool closed on April 1, with bellwethers to be selected in the coming months. The parties continue to work through general discovery issues, and depositions are set to begin in August of this year.
March 8, 2024: Court Rejects Defendants' Bid to Restrict Discovery
The Court held a case management conference on March 1, 2024, during which the Defendants argued to severely restrict the discovery that the Plaintiffs may conduct. The Defendants were unsuccessful. The parties will meet with the Court again on March 29, 2024, to discuss the progress of discovery.
February 2, 2024: Discovery Progress and Preservation Order Discussed in Phoenix
Discovery is ongoing, and the parties are due back in court in Phoenix on March 1, 2024. At that time, the parties will report their progress in discovery and raise any discovery disagreements with Judge Campbell. The judge has also asked that the parties finalize a preservation order that would apply to port devices that are explanted because of injury.
The Road to Justice for Bard PowerPort Victims: Litigation Efforts in 2023 and Prior
- November 16, 2023: Judge Campbell held a status conference in his courtroom in Phoenix, Arizona, after which he entered a Master Complaint and a Short-Form Complaint, allowing Plaintiffs to file their case directly into the MDL. He also issued a case management order setting deadlines for the case. The discovery period, during which Plaintiffs can gather evidence, will run from November 20, 2023, to January 31, 2025. Following this period, the parties will submit expert reports, and trials are expected to begin in 2026.
- October 18, 2023: Judge Campbell has ordered the parties to cooperate in filing proposed protective orders, electronic discovery orders, and preservation orders by October 27, 2023. He also ordered the parties to meet and confer regarding the form of complaints and direct filing orders, among other preliminary issues. According to attorney Rebecca Phillips, “We will work with the Defendants’ counsel to help the Court get the MDL up and running as efficiently as possible as soon as possible.” The next hearing will occur in Phoenix on November 16, 2023.
- September 19, 2023: The US District Court for the District of Arizona has appointed Lanier Law Firm’s own Rebecca Phillips as Co-Lead Counsel on the Plaintiff’s Leadership Counsel on MDL No. 3081, In Re: Bard Implanted Catheter Product Liability Litigation.
- August 9, 2023: Yesterday, the Judicial Panel on Multidistrict Litigation centralized federal PowerPort cases in the United States District Court for the District of Arizona before the Honorable David Campbell.
- July 27, 2023: Plaintiffs argued before the Judicial Panel on Multidistrict Litigation (“JPML”) that PowerPort cases should be consolidated for pretrial proceedings in the Western District of Missouri. Defendant opposed consolidation and proposed either Utah or Arizona as appropriate venues if the JPML was inclined to consolidate the cases.
- June 16, 2023: The United States Judicial Panel on Multidistrict Litigation filed a notice that it will hear oral arguments on July 27, 2023, in San Francisco about whether to create a centralized federal MDL.
- May 24, 2023: Plaintiffs in various cases requested that the United States Judicial Panel on Multidistrict Litigation consolidate Bard PowerPort Lawsuits into a multidistrict litigation in the Western District of Missouri.
- July 8, 2019: The FDA cleared Bard Power-Injectable Implantable PowerPorts through the 510(k) process.
- June 7, 2019: The FDA makes the ASR database public. In response to the public outcry to the Kaiser Health report, and starts requiring companies to report adverse events through the publicly accessible MAUDE database.
- March 7, 2019: Kaiser Health publishes a report revealing that medical device companies were filing reports of device malfunctions to the FDA in secret.
- December 29, 2017: Becton Dickinson and Co. acquires C.R. Bard and is believed to have taken on Bard’s liabilities.
- February 14, 2014: The FDA clears the PowerPort Implantable Port through the 510(k) process.
- November 15, 2012: The FDA clears the PowerPort Clearvue Slim Implantable Port through the 510(k) process.
- March 27, 2009: C.R. Bard receives a 510(k) clearance for the PowerPort Due MRI Implanted Port with 9.5 Fr. Dual Lumen Chronoflex Polyurethane Catheter.
- June 4, 2008: The FDA clears the Powerport Implanted Port with Groshong Catheter.
- December 19, 2007: C.R. Bard receives a 510(k) clearance for the MRI Powerport Implanted Port with 9.6 Fr. Silicone Catheter.
- November 14, 2007: C.R. Bard receives clearance through the FDA’s 510(k) process for the Titanium PowerPort Isp Implanted Port with 6 Fr Chronoflex Polyurethane Catheter.
- November 1, 2007: C.R. Bard receives clearance through the FDA’s 510(k) process for the Titanium PowerPort Isp Implanted Port with 6 Fr Chronoflex Polyurethane Catheter.
- January 25, 2007: C.R. Bard receives an FDA 510(k) clearance for the Powerport Polymetric Port with 8 Fr S/L Chronoflex Catheter.
- July 14, 2006: The FDA grants multiple 510(k) clearances for C.R. Bard’s Implanted Titanium Port with 8 Fr. Chronoflex Catheter.
Call 800-723-3216 now to see if our law firm can pursue compensation for you and your family. Consultations are free.
Why Choose the Lanier Law Firm to Handle Your Bard PowerPort Lawsuit?
Since our establishment in 1990, our nationally recognized defective products lawyers have won nearly $20 billion in verdicts and settlements on behalf of our deserving clients, including the following:
$9 billion
verdict against Takeda & Eli Lilly for cancer associated with the diabetes drug Actos
$4.85 billion
National Vioxx Settlement against Merck & Co.
$4.69 billion
verdict against Johnson & Johnson in the first trial linking asbestos in baby powder to ovarian cancer
Going against large drug companies like Becton Dickinson and Bard requires extensive financial resources that can be invested upfront. It also requires trial skills that can match the skills of the most expensive lawyers hired by the pharmaceutical companies.
At the Lanier Law Firm, we are not afraid to go against large companies. Law360 designated us the Most Feared Plaintiffs Firm in 2014. Our attorneys have also received prestigious awards, including the following:
- Best Lawyers in America
- Most Impressive Plaintiff Verdict by Courtroom View Network
- Best Law Firms, Tier 1 Ranking
- Largest Verdict of 2018, by Verdict Search and the National Law Journal
Our founder, Mark Lanier, has been recognized by the National Trial Lawyers as the 2018 Trial Lawyer of the Year and is included among their top 50 most influential lawyers in the United States.
What Is a Bard PowerPort?
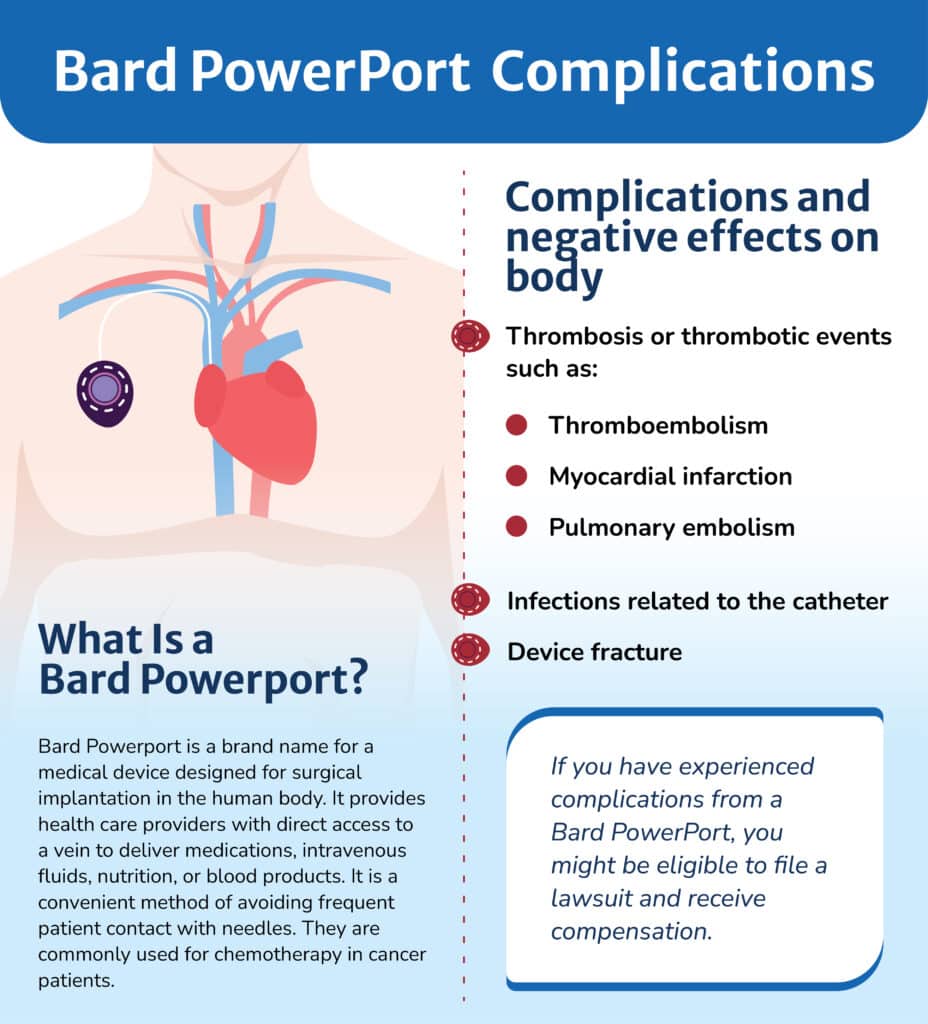
Bard Powerport is a brand name for a medical device designed for surgical implantation in the human body. It provides health care providers with direct access to a vein to deliver medications, intravenous fluids, nutrition, or blood products. It is a convenient method of avoiding frequent patient contact with needles. They are commonly used for chemotherapy in cancer patients.
What Is the Problem with Bard PowerPorts?
The catheter is made from a type of polyurethane and silicone. It contains barium sulfate, a contrast dye that makes the port more visible during imaging tests, such as X-rays and CT scans. Unfortunately, barium sulfate has been shown to degrade polyurethane and silicone when in contact with human tissue.
Get a Free Legal Consultation
- Passionate legal advocates with decades of trial experience
- Over $20 billion recovered for our clients
- No upfront costs or fees
The Effects and Complications Associated with Bard PowerPort Catheters
The complaints also allege that catheter particles enter the bloodstream and travel to various internal organs, causing organ damage and life-threatening infections. According to complaints, patients with the implant have an increased risk of the following:
- Thrombosis or thrombotic events such as:
– Thromboembolism – A life-threatening blood clot that blocks blood flow in the veins
– Myocardial infarction symptoms – Heart attack symptoms, such as chest pain, dizziness, and difficulty breathing
– Pulmonary embolism – a blood clot that has traveled to the lungs, which blocks blood flow to the lungs and can be deadly without emergency treatment - Infections related to the catheter
- Device fracture
Additionally, some plaintiffs also allege that they have experienced severe or persistent pain, tearing of internal body structures, hematomas, cardiac arrhythmia, and fluid buildup around the heart. Some have required surgery.
Complaints allege that the particles of barium sulfate break down from the surface of the PowerPort over time, leaving divots, pits, cracks, microfractures, and breakage of the device. The device may malfunction or migrate to other parts of the body.
Liability for Defective PowerPort Catheter Defects
Manufacturers of medical devices are required to ensure that the products they make available to the public are safe and effective.

What Did Bard Know About the PowerPort’s Defects?
According to the pending lawsuits, Bard received a large number of adverse event reports from health care providers soon after introducing the PowerPort to the market, indicating that the company knew or should have known that patients implanted with its PowerPorts had increased risks of potentially life-threatening injuries.
Complaint allege that when C.R. Bard began receiving adverse event reports, it may have prevented hundreds of injuries and deaths by doing even one of the following:
- Recall the product
- Remove the barium sulfate from the product
- Encapsulate the barium sulfate
- Use a different polymer formulation
Update the warning label to disclose the known risks
Instead, Bard continues to market the product as safe and effective.
Complaints allege that not only did the company fail to take steps to protect the public, but it may have deliberately concealed the product’s harmful effects from the public.
Who Is Liable for Harm Caused by Bard PowerPort Catheters?
Becton Dickinson acquired Bard in 2017 as a wholly-owned subsidiary. As a result, Becton Dickinson and Bard may be liable.

When Should I Contact a Bard PowerPort Attorney?
Every state sets its own deadline for filing a lawsuit, known as the statute of limitations. If you do not file before the deadline expires or if your case is not tolled, you can lose your right to pursue justice for your injuries. For this reason, it is imperative that you contact an attorney as soon as possible after you discover your injuries.
Our hard-working and dedicated product liability attorneys can determine how the statute of limitations affects your case and ensure your case is filed on time. Reach out to our talented legal team at 800-723-3216 or fill out our contact form to start your claim today.

Rebecca Phillips is dedicated to pharmaceutical liability, business litigation, and appeals. Co-lead counsel in the Bard Implant litigation, she also helped secure billions in opioid recovery for Texas. Recent honors include Super Lawyers Rising Star and Lawdragon 500 (2024-2025), among others.
Contact the Lanier Law Firm to Start Your Claim Today
If you or your loved one received any of the following Bard PowerPorts and suffered an injury due to catheter migration or fracture, including infections, organ damage, and life-threatening blood clots, you may have a case against Bard and Becton Dickinson:
- Bard Power-Injectable Implantable Ports (Powerports®)
- Bardport®, Slimport®, And X-Port® Implanted Ports
- Power-Injectable Implantable Ports With Chronoflex Polyurethane Catheters
- Powerport Implantable Port
- Powerport Clearvue Slim Implantable Port
- Titanium Powerport Isp Implanted Port
- Powerport Duo MRI Implanted Port With 9.5 Fr. Dual Lumen Chronoflex Polyurethane Catheter
- Powerport Implanted Port With Groshong Catheter
- MRI Powerport Implanted Port With 9.6 Fr Silicone Catheter
- Titanium Powerport Isp Implanted Port With 6 Fr Chronoflex Polyurethane Catheter
We have a winning record against drug companies and medical device manufacturers. Contact us today for a free case evaluation.
Bard Port Lawsuit FAQs
Below are answers to common questions about Bard PowerPort lawsuits.
How Were Such Dangerous Products Cleared by the FDA?
Bard PowerPorts were cleared through the FDA’s Premarket Notification 510(k) program, which allows manufacturers to receive clearance for products that are, in theory, substantially similar to existing products without having to complete clinical studies verifying a product’s safety and efficacy.
Is the Bard PowerPort Device Recalled?
There is currently no active recall for these products. Some Bard PowerPorts were recalled in October 2019 for unrelated reasons, but this recall was terminated February 18, 2022.
Is There a Class-Action Bard PowerPort Lawsuit?
There is no class-action lawsuit, but a motion to consolidate the cases into multidistrict litigation, or MDL, has been filed.
When cases are transferred to an MDL, case discovery is shared, but each case remains separate. A few representative cases are usually selected to go to trial as a test to determine the likely outcome for the other cases. These are known as bellwether trials. These trials can provide guidance for how the remaining cases should resolve. Our mass tort lawyers have extensive experience with these types of cases and have served in leadership roles in large MDLs.
How Much Does a PowerPort Attorney Cost?
At The Lanier Law Firm, the initial consultation is free, and we work on a contingency fee arrangement. This means that we take on all the financial risks associated with your case, investing our own resources into filing fees, expert witnesses, and other case costs, with nothing due from you until after we recover compensation for you.























