DePuy/J&J Hip Implant Litigation
Home » Practice Areas » Pharmaceutical Liability » DePuy/J&J Hip Implant Litigation
Medical devices are meant to improve longevity and quality of life. Doctors and patients rely on medical device manufacturers to provide safe, effective products with full disclosure of the risks and benefits so that doctors can deliver quality care and patients can make informed decisions.
The Food and Drug Administration (FDA) receives hundreds of thousands of reports every year of deaths, serious injuries and malfunctions concerning medical devices. Manufacturers that provide unsafe products to the public are liable when patients are harmed.
Johnson & Johnson and its subsidiary, DePuy Orthopaedics, have a long history of harming patients through their defective hip implants. The pharmaceutical liability attorneys at The Lanier Law Firm stand up to large pharmaceutical companies on behalf of innocent individuals.
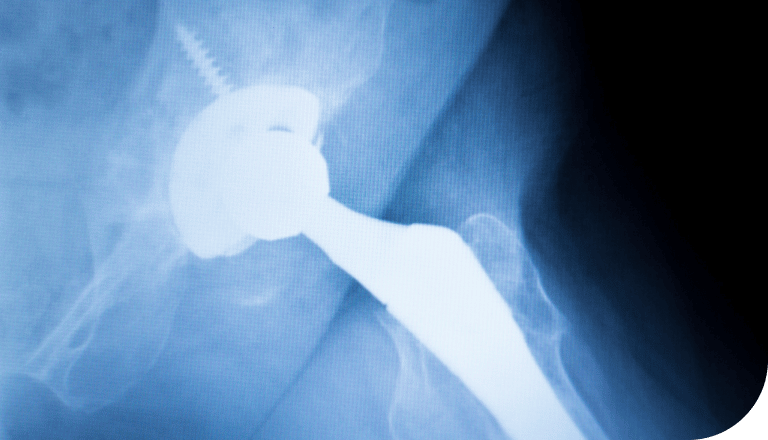
Harm Caused by DePuy’s Hip Implants
- Metal particles entering the surrounding tissues, resulting in the following:
- Deterioration of the tissue
- Loosening of the implant
- Failure of the device
- Metal ions entering the bloodstream and impacting all parts of the body and brain as follows:
- Skin rash
- Cardiomyopathy
- Neurological changes, including changes in hearing or vision
- Psychological changes, including depression or cognitive impairment
- Renal function impairment
- Thyroid dysfunction
DePuy/J&J marketed and sold a product it knew could cause these problems without warning the public. Below are just a few examples of the ill effects our clients experienced:
- Severe pain and inflammation
- Bone erosion
- Tissue loss
- Painful revision surgeries
Deceptive Marketing Practices
DePuy/J&J knew its products were harmful, but rather than develop a safer product, the company heavily marketed the product and deliberately withheld information from doctors and patients about the risks associated with using its products.
By submitting this form, you agree to our terms & conditions. Please read full disclaimer here.
Harm Caused by DePuy’s Hip Implants
The Lanier Law Firm’s Successful Litigation History Against Johnson & Johnson and DePuy/J&J
How are defective medical device cases proven?
Who is liable for defective medical devices?
What is the difference between a class-action lawsuit and MDL?
How to Choose a Defective Medical Device Lawyer
Contact the Experienced Legal Team at The Lanier Law Firm Today
Fraud
DePuy/J&J failed to provide adequate warnings about its products’ risks and went to such lengths to conceal this information that one verdict found that this rose to the level of fraud. The court also determined the fraud was aided and abetted by the parent company, Johnson & Johnson.
Fraudulent Marketing Techniques
The aggressive marketing tactics employed by Johnson & Johnson and DePuy/J&J included the following:
- Offering kickbacks to surgeons for using the device, even though they knew of safer alternatives
- Intimidation of at least one doctor who intended to testify against them, according to a Law360 report

The Lanier Law Firm’s Successful Litigation History Against Johnson & Johnson and DePuy/J&J
2010 Recall of ASR-Branded Products
In August of 2010, Johnson & Johnson announced that two hip replacement products, the ASR XL Acetabular System and the ASR Hip Resurfacing System, were recalled after more than 10 percent of patients required corrective surgeries within five years of their initial hip replacement procedures. More than 93,000 patients had purchased these products, netting over $5 billion in sales in 2009 alone.
Due to the high volume of cases, the cases were consolidated into multidistrict litigation (MDL). The case was In re: DePuy/J&J Orthopaedics Inc. ASR Hip Implant Products Liability Litigation (MDL 2197). The Houston and Los Angeles offices of The Lanier Law Firm provided national coordination for the case and represented 100 clients across 30 states and overseas.
2011 MDL Against DePuy/J&J
Thousands of lawsuits regarding DePuy/J&J’s Pinnacle hip implants were consolidated in the U.S. District Court for the Northern District of Texas. The case is In Re: DePuy/J&J Orthopaedics, Inc., Pinnacle Hip Implant Products Liability Litigation (MDL 2244).
2013 Removal of Hip Liners from the Market
These products had been sold from 2002 to 2012 until DePuy/J&J pulled the metal liners off the market in 2013. The hip implant systems were never recalled.
2016 Bellwether Verdicts in the Pinnacle Implants MDL
In two of the early bellwether trials, Texas juries awarded $502 million to our clients from Texas and more than $1 billion to our clients from California. Law360 reported that these awards included pain and suffering damages and loss of consortium for four spouses.
These were later reduced to $150 million and $543 million. Mark Lanier, founder of The Lanier Law Firm, responded to the verdict as follows:
“Once again, a jury has listened to the testimony of both sides and returned a verdict affirming what we’ve known all along: a responsible company would settle these cases and take care of their injured consumers, rather than forcing them through expensive and vexatious litigation just to delay justice. This jury spoke loud and clear, and I hope J&J will finally listen.”
In addition to Mark Lanier, the members of the trial team included the following attorneys from The Lanier Law Firm:
- Alex J. Brown
- Michelle Carreras
- Dr. Robert Leone
- Kevin P. Parker
The firm also collaborated with two other law firms, including Ernest Cannon Law.
2017 Bellwether Verdicts in the Pinnacle Implants MDL
A Texas federal jury unanimously awarded a $247 million combined verdict in favor of six New York plaintiffs against Johnson & Johnson and DePuy/J&J for manufacturing defects, fraud and deceptive trade practices.
This award included $90 million in punitive damages against Johnson & Johnson and $78 million in punitive damages against DePuy/J&J. The verdict was upheld in 2018, with a final combined judgment of $245 million.
Case Settlements
Despite the overwhelming evidence against them, Johnson & Johnson and DePuy/J&J resisted settling claims and instead subjected the patients they had harmed to years of stressful litigation. Thanks to the large jury verdicts, they finally realized settling was in their best interest, though they have yet to admit wrongdoing.
2013 ASR Settlement
Bloomberg reported that Johnson & Johnson and DePuy/J&J settled the ASR Implants cases in 2013 for $4 billion (including legal fees) in the United States. This is in addition to similar cases the companies settled in India.
2019 Pinnacle Settlement
The Bloomberg report also revealed that Johnson & Johnson agreed to pay approximately $1 billion to settle more than 95 percent of the 6,000 cases involving the Pinnacle Ultamet devices in which doctors had to remove the implants. The Lanier Law Firm played a leading role in negotiating this settlement.
How are defective medical device cases proven?
Manufacturers of defective medical devices are liable when their devices cause harm and one or more of the following can be proven:
- The product’s design is harmful.
- A product is defective due to a manufacturing error or quality control deficiency.
- The manufacturer fails to provide adequate warnings or instructions.
In the case of DePuy/J&J, two of the three criteria applied: the products were inherently harmful, and the company failed to provide adequate warnings about the risks.

Who is liable for defective medical devices?
Anyone in the chain of distribution, from the manufacturer to the health care provider, could be liable, including but not limited to the following:
- Manufacturers
- Marketers, including sales representatives who knowingly misrepresent product safety
- Distributors
- Health care providers who fail to provide safety information or improperly implant the devices
- Retailers
What is a mass tort?
A mass tort occurs when a large number of litigants have similar case facts pertaining to the same products. They are common in product liability cases, especially pharmaceutical liability cases. In an effort to provide efficient management of these cases and prevent them from overwhelming the court system, they may be consolidated into MDL or class-action cases.
What is the difference between a class-action lawsuit and MDL?
In class-action lawsuits, all the cases are combined into one case. Any resulting award is divided between the class members.
In MDL, each case remains separate, but all the cases are assigned to one federal court. The court will select a few representative cases, known as bellwether cases, as test cases to determine the likely outcome in the remaining cases. This often decreases the number of cases that must be heard because the remaining cases are more likely to settle or be dismissed.
How to Choose a Defective Medical Device Lawyer
When selecting an attorney, it is important to keep in mind that medical device cases require the highest level of legal skill due to their complex nature. Medical device manufacturers are generally companies with billions of dollars in resources, and they fight vigorously to avoid liability.
Experience
Experience is of the utmost importance when selecting a pharmaceutical liability attorney. Medical device manufacturers capitalize on even the smallest mistakes, and they will not shrink from using every legal maneuver in the book to avoid paying the compensation they rightfully owe.
These companies have consistently demonstrated that they are willing to spend more on litigation than the amount they owe the patients wronged. An inexperienced or incompetent attorney cannot fight these companies effectively.
Our attorneys have helped clients who were harmed by the following medical devices, in addition to hip implants:
- C.R. Bard transvaginal mesh
- Philips CPAP and ventilator machines
- Medtronic Sprint Fidelis lead wires
- Mirena intrauterine device (IUD)
- Bard Composix Kugel mesh patch
- Numerous dangerous drugs
We have been standing up to drug companies for more than 30 years. We are familiar with their tactics, and even their highest-paid attorneys are no match for the skilled attorneys at The Lanier Law Firm.
National Reputation
A law firm that is well-known and respected by the pharmaceutical companies has the ability to get better results than lesser-known attorneys who have yet to prove their prowess. The Lanier Law Firm has been named among one of Law360’s Most Feared Plaintiffs’ Firms because it has a history of obtaining very high verdicts.
Track Record
One of the most important measures of a law firm’s caliber is its history of results. A law firm’s track record demonstrates the type of results you can expect to receive in your case.
In addition to the billions of dollars in verdicts and settlements we have achieved against DePuy/J&J, The Lanier Law Firm has won significant awards on behalf of very deserving clients who were harmed by defective medical devices, including the following:
- $7.25 billion total verdicts against C.R. Bard for transvaginal mesh defects
- $56 million verdict against Biomet for defective hip implants
- $1.2 billion nationwide settlement against Sulzer Orthopedics for defective Inter-Op brand hip replacements
Richard Meadow is one of the nation’s foremost pharmaceutical liability attorneys, with prestigious recognition by his peers as a leader in the industry and honors including Texas Super Lawyer, Law Dragon’s Top 500 Lawyers in the Country and placement on U.S. News & World Report’s Best Lawyers list.
In addition to his role in the DePuy/J&J cases, Mr. Meadow has played significant roles in achieving substantial verdicts and settlements in cases involving the following defective medical devices:
- Zimmer Duron hip cups
- Zimmer Nexgen knee implants
- American Medical Systems, Inc.’s pelvic repair systems
- Boston Scientific Corp.’s pelvic repair system
- Mirena IUD
Resources
Pursuing pharmaceutical companies requires vast resources, which small, local law firms often lack. The Lanier Law Firm has locations in Texas, California and New York, and we have the ability to travel anywhere in the country at a moment’s notice. We have a staff of more than 60 attorneys with impressive litigation experience, and our network includes some of the foremost medical experts and forensic scientists in the industry.
Mark Lanier is well-known for his television-worthy courtroom performances and his use of visual aids in the courtroom. He has paid thousands of dollars for obscure products on eBay to prove his cases. Our attorneys not only have access to resources, but they are resourceful.
Contact the Experienced Legal Team at The Lanier Law Firm Today
Our philosophy at The Lanier Law Firm is redefining legal care. To our lawyers, legal care is providing injured individuals with the highest caliber of legal representation available so they stand a fair chance against companies with massive resources.
If you have been harmed or lost a loved one because of a defective medical device, you may be able to recover substantial compensation. We use a contingency fee arrangement, so you will not have to pay any legal fees unless and until we recover compensation for you.
This means you have nothing to lose by speaking with an attorney, but your jurisdiction may limit the amount of time you have to pursue your case. It is important to schedule a case review as soon as possible after your injury. Contact us today to schedule a free consultation.
By submitting this form, you agree to our terms & conditions. Please read the full disclaimer



